|
1/13/2024 0 Comments Modern periodic table with details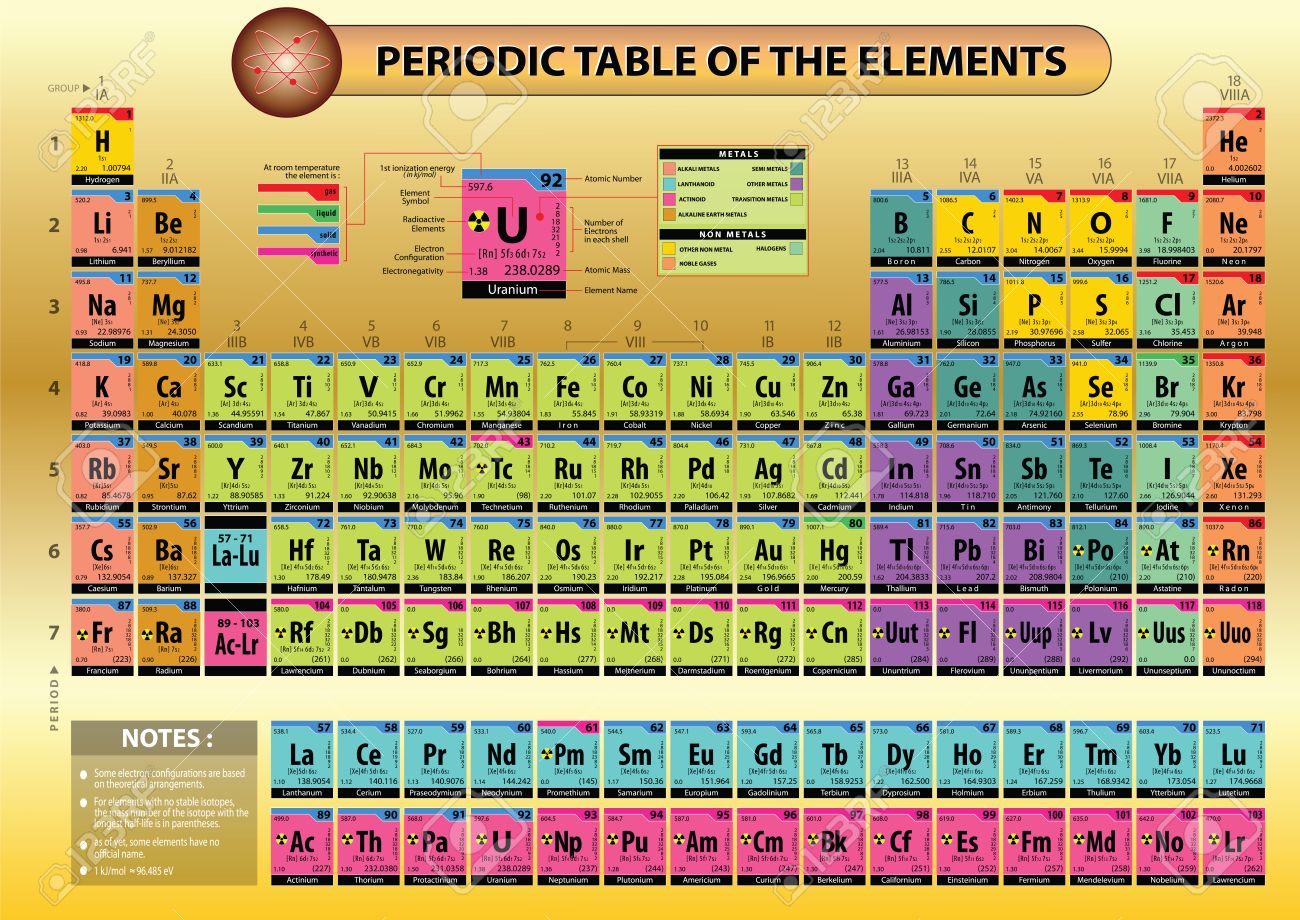
An element's period number is the highest unexcited energy level for an electron of that element. The rows of the periodic table are called periods.At the time of this writing, the most recent version of the periodic table was approved in December 2018. The International Union of Pure Applied Chemistry, IUPAC, revises the periodic table as new data becomes available.It is the lightest element that has only radioactive isotopes (none are stable). Technetium was the first element to be made artificially.Some sources state more elements occur naturally because heavy elements may transition between elements as they undergo radioactive decay. All of the other elements are strictly human-made. There are about 94 elements on the periodic table that occur in nature.It wasn't the first table that organized the elements according to periodic properties. Although Dmitri Mendeleev is most often cited as the inventor of the modern periodic table, his table was just the first to gain scientific credibility.Retrieved from Ĭopyright © Israel Science and Technology Directory. "Sortable list of elements of the Periodic Table". The story behind the discovery that elements are born in stars.Atomic Weights of the Elements (From IUPAC).Multilingual Dictionary and Etymology of the Periodic Table Elements.Atomic Reference Data for Electronic Structure Calculations.List of Periodic Table Elements in Hebrew.Other resources related to the Periodic Table For these elements, the weight value represents the mass number of the longest-lived isotope of the element.Įlectron configuration: See next page for explanation of electron configuration of atoms. The elements marked with an asterisk have no stable nuclides. The values shown here are based on the IUPAC Commission determinations ( Pure Appl. For relative abundances of isotopes in nature, see reference on Atomic Weights and Isotopic Compositions.Ītomic weight: Atomic weight values represent weighted average of the masses of all naturally occurring isotopes of an element. The abundance of each isotope depends on the source of materials. For example, the two common isotopes of carbon, 12C and 13C, have 6 and 7 neutrons, respectively. 
Elements have more than one isotope with varying numbers of neutrons. The isotope of an element is defined by the sum of the number of protons and neutrons in its nucleus. Isotope: Atoms of the same element with the same atomic number, but a different number of neutrons. Thus, each proton and neutron has a mass of about 1 amu. This isotope of carbon has 6 protons and 6 neutrons. Atomic mass is measured in Atomic Mass Units (amu), which are scaled relative to carbon, 12C, that is taken as a standard element with an atomic mass of 12. 
Each element is uniquely defined by its atomic number.Ītomic mass: The mass of an atom is primarily determined by the number of protons and neutrons in its nucleus. Boiling pointĪtomic number: The number of protons in an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
